 
We will process your data until the communication with you is complete or until you object, unless the law obliges us to process it for a longer period or in case of potential claims, we will store it for the duration of the limitation period which is determined by law, in particular the Civil Code. We process your data in order to send you a newsletter - the basis for processing is the implementation of our and third parties' legitimate interests - direct marketing of our products / products of the PCC Group. Of course, just like any other acidic food, H 2CO 3 consumed in excess can cause disruption to the acid–base balance in the body. So, what is the truth, and who is right? Carbonated water or beverages usually have a slightly acidic taste, and although they can cause distressing symptoms, these are only relevant for those who struggle with gastrointestinal problems or indigestion. Some claim that the consumption of carbonic acid beverages contributes to the acidification and wasting of the stomach, whereas others believe that H 2CO 3 has no effect whatsoever on the human body. There are many myths surrounding carbonic acid. How does carbonic acid affect the human body? In medicine, this compound is used, for example, by health resorts to prepare carbonic baths (recommended mainly for hypertensive patients and patients suffering from cardiovascular diseases). It is safe to say that without this ingredient, we would never have known the taste of iconic beverages, such as classic soda water, carbonated mineral water or soft fizzy drinks such as orangeade, Coca-Cola, Pepsi or Sprite.Ĭarbonic acid also finds application in laboratories in the chemical industry, as well as in cosmetology – it is used to prepare advanced skin cleansing and rejuvenating treatments. CO 2 was obtained as a result of the reaction of acetic acid with sodium bicarbonate (baking soda). After saturating water with it, it was found that a carbonated beverage is much more refreshing than any other known soft drink.Ĭurrently, the range of application of carbonic acid (H 2CO 3) in the carbonated beverages industry is very broad. This property was used more than 200 years ago by the first soda makers who came up with the idea of passing carbon dioxide through water. Occurrence and application of carbonic acidĬarbonic acid is distinguished by the fact that it occurs only in aqueous solutions. As a result of the dissociation, the entire chemical is broken down into 2 positive hydrogen ions and negative carbonate ions. It is a two-step process which consists of gradual disconnection of hydrogen ions – first, one cation falls off, and then another. This phenomenon is called ionic dissociation. What is carbonic acid dissociation?Ĭarbonic acid in aqueous solutions is gradually decomposed. 
Contemporary scientists have also found that carbonic acid can be obtained in gaseous form in trace amounts, although this is a very difficult task. Furthermore, carbonic acid cannot be produced in a concentrated form. 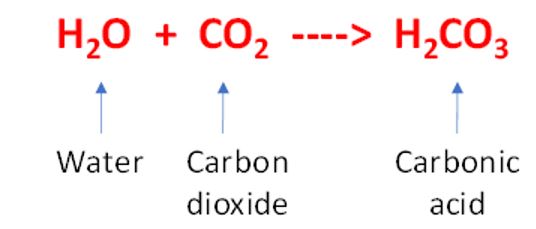
co-creates the salts of carbonic acid (carbonates).ĭibasic acid H 2CO 3 forms two types of salts: acidic (bicarbonates) and neutral ones.is readily degradable to water and carbon dioxide,.What should you know about the physical and chemical properties of carbonic acid? This compound: The chemical obtained during this reaction shows several specific properties, which we will discuss below. How is H 2CO 3, or carbonic acid, produced? The structural formula of this chemical indicates that it is formed through the dissolution of carbon dioxide (CO 2) in water (H 2O). It also does not require too many ingredients or the use of advanced technologies. The production process of carbonic acid is not complicated. Production and decomposition of carbonic acid 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
